These molecules form the basis for life and are studied in great detail in the chemistry disciplines of organic chemistry and biochemistry.
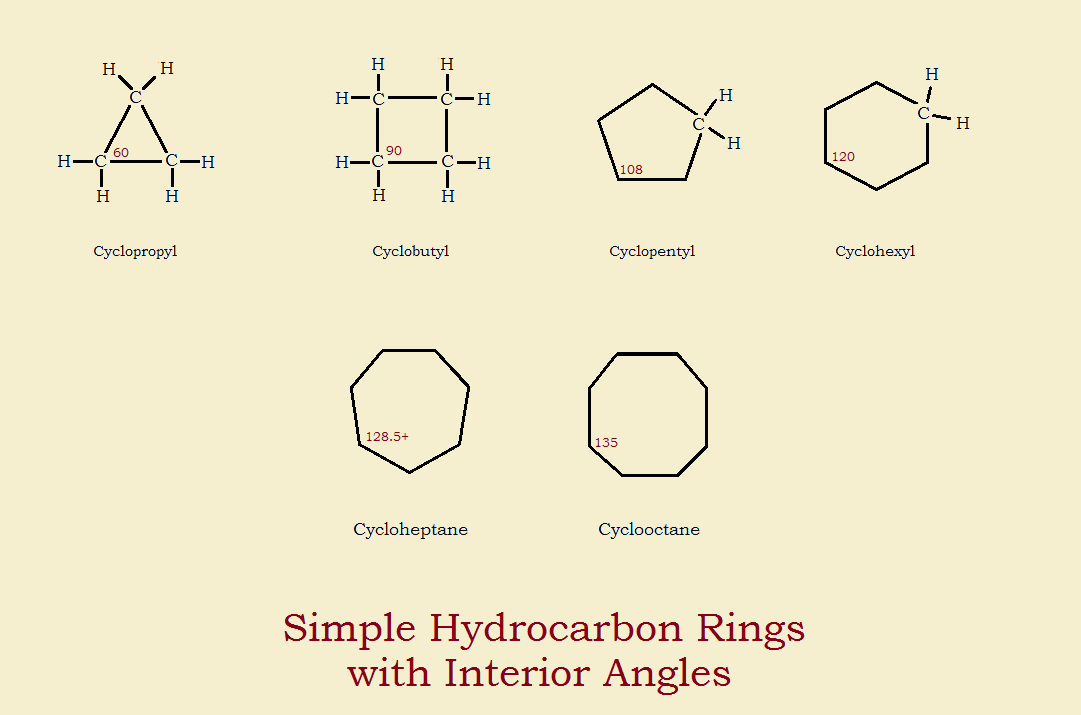
Types of rings in organic chemistry.
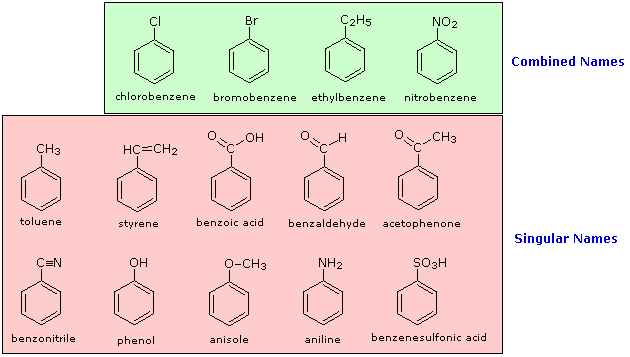
As organic chemistry grew and developed many compounds were given trivial names which are now commonly used and recognized.
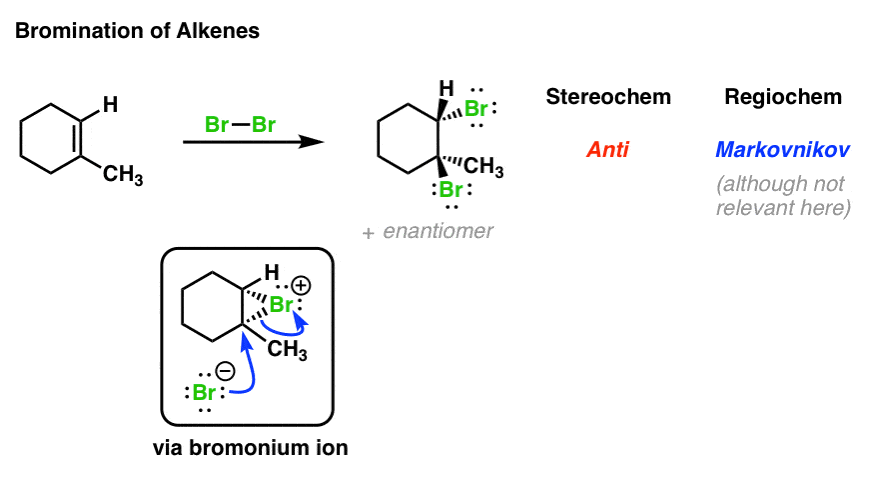
The primary goal of this lesson will be to understand a phenomenon in organic chemistry known as ring flip by studying its definition and looking at specific example structures to illustrate the.
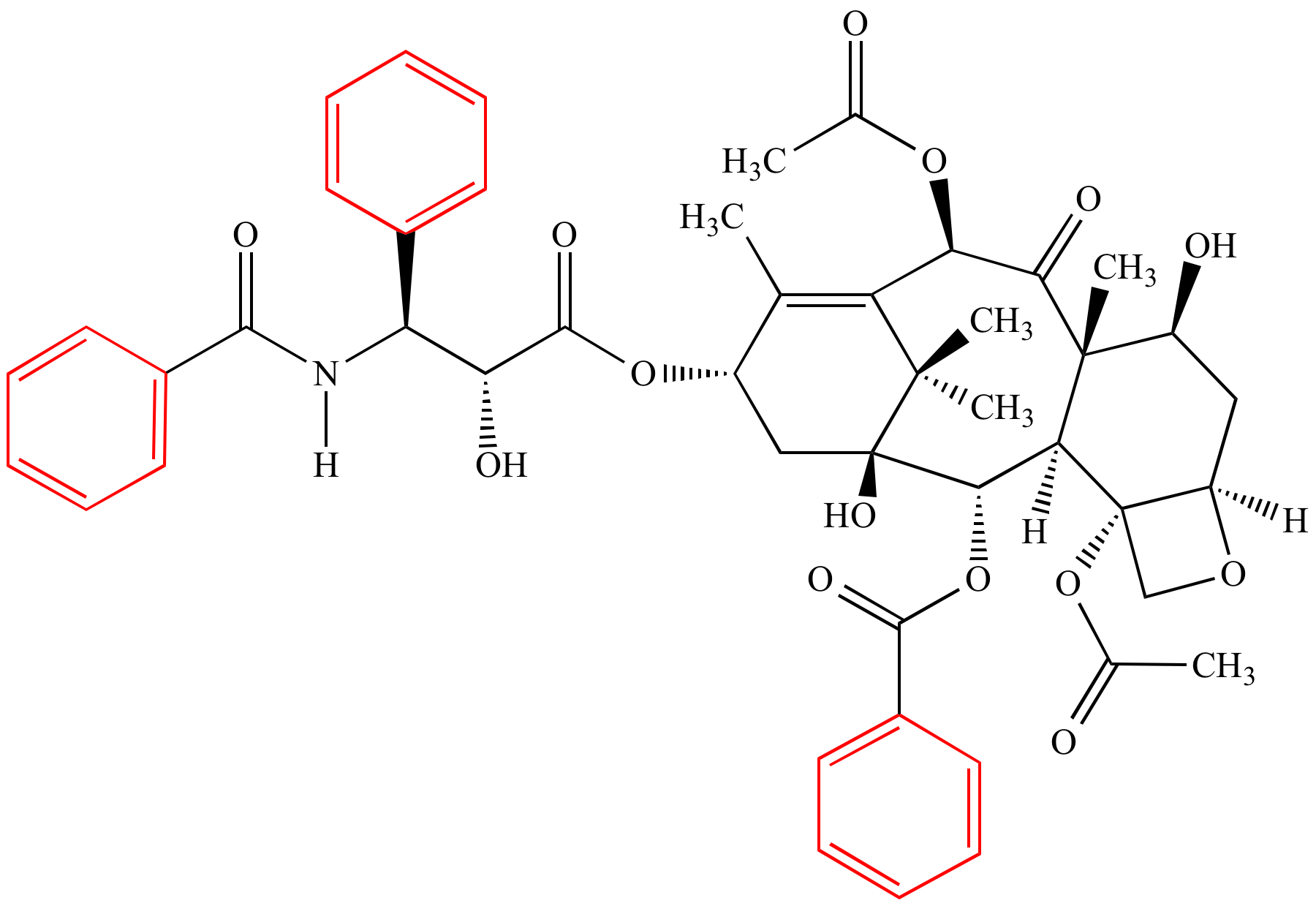
It is also a comprehensive summary of the many techniques and reagents used in total synthesis.
This set of pages is a collection of short natural product syntheses and a few unnatural products.
Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis properties and applications of these heterocycles.
Currently about 700 syntheses are included and indexed by compound names types of reactions reagents named reactions used chemoselectivity number of rings and years published.
A cyclic compound ring compound is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring rings may vary in size from three to many atoms and include examples where all the atoms are carbon i e are carbocycles none of the atoms are carbon inorganic cyclic compounds or where both carbon and non carbon atoms are.
There are four main types or classes of organic compounds found in all living things.
Many organic compounds contain rings of carbon atoms or other atoms such as oxygen or nitrogen.
The simplest ring compound contains 3 carbons as in cyclopropane.
The molecular formula of a hydrocarbon provides information about the possible structural types it may represent.
Cycloalkanes have one or more rings of carbon atoms.
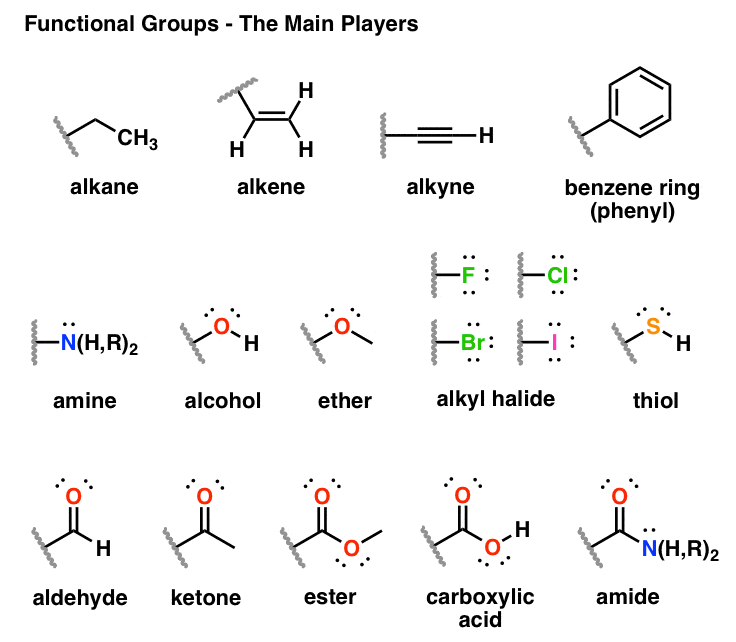
Functional groups undergo the same chemical reactions no matter how large or small the molecule is.
There are many different types of strain that contribute to the overall ring strain in cycloalkanes including angle strain torsional strain and steric strain.
Small rings such as three and.
Baeyer suggested that cyclopropane and cyclobutane are less stable than cyclohexane because the the smaller rings are more strained.
In 1890 the famous german organic chemist a.
Examples of heterocyclic compounds include all of the nucleic acids the majority of drugs most biomass cellulose.
The most common ring compounds contain either 5 or 6 carbons.
Carbohydrates lipids proteins and nucleic acids.
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring s.
These compounds are known as cycloalkanes.
Organic compounds are called organic because they are associated with living organisms.
Many organic compounds found in nature contain rings of carbon atoms.